Refining
Topics
Refined Products and Qualities
Crude Oil Fractions
Crude oil is processed or refined to produce useable products such as gasoline. The process is very complex and involves both chemical reactions and physical separations. Crude oil is composed of thousands of different molecules. It would be nearly impossible to isolate every molecule and make finished products from each molecule. Chemists and engineers deal with this problem by isolating mixtures of molecules according to the mixture's boiling point range. For example, gasoline molecules might boil in the range from 90 to 400 oF. Home heating oil could be from molecular mixes that boil from 500 to 650 oF. For convenience, the mixtures or fractions are given a name. The following chart illustrates the boiling range and name of the petroleum fraction.
| Fraction | Boiling Range, oF. |
| Butanes and lighter | <90 |
| Light straight run gasoline (LSR) or light naphtha (LN) | 90-190 |
| Naphtha or heavy naphtha (HN) | 190-380 |
| Kerosene | 380-520 |
| Distillate or atmospheric gas oil (AGO) | 520-650 |
| Residua | 650 + |
| Vacuum gas oil (VGO) | 650-1000 |
| Vacuum Residua | 1000 + |
Refined products are produced by combining fractions from the raw crude oil with those from various refinery processing units. These fractions are mixed or blended to satisfy specific properties that are important in allowing the refined product to perform as desired in an engine, for ease in handling and to reduce the undesirable emissions produced when the product is burned.
Product Specifications
Gasoline is blended to meet the following specifications:
| | Reid Vapor Pressure (RVP) which is a measure of hydrocarbon vapors and is needed for starting engines. |
| | Octane which is a measure of anti-knock level of gasoline and is important because knocking lowers engine efficiency and wastes power. |
| | Toxics which are measures of the harmful components in gasoline and refiners are required to benzene, olefins and sulfur levels. |
| | Oxygen content in reformulated gasolines to reduce the level of green house gas emissions. |
Jet fuel is blended to meet the following specifications:
| | Freeze Point is the temperature at which the fuel forms ice crystals which could clog engine fuel filters. |
| | Viscosity is a measure of how easily the jet fuel flows. |
Diesel engines are different than gasoline engines, and, as result, have different specifications:
| | Cetane Index is a measure of engine performance. |
| | Sulfur content determines the level of sulfur oxides in the exhaust. |
| | Pour Point is the temperature at which the diesel fuel flows. |
| | Viscosity is a measure of how easily the diesel fuel flows. |
Refinery Operations
Crude Oil Distillation
Crude oil distillation is used to separate the hydrocarbons in crude oil into fractions based on their boiling points. The separation is done in a large tower that is operated at atmospheric pressure. The tower contains a number of trays where hydrocarbon gases and liquids interact. The liquids flow down the tower and the gases up. The lighter materials such as butane and naphtha are removed in the upper section of the tower and the heavier materials such as distillate and residual fuel oil are withdrawn from the lower section.
Vacuum Distillation
The residua fraction (650 oF. and higher boiling material) from the crude tower can be sent to fuel blending to produce residual fuel oil or No. 6 fuel oil. Often this residua fraction is further separated into a vacuum gas oil and vacuum residua. This unit is operated at a slight vacuum. This allows the hydrocarbons to be separated at lower temperatures and prevent undesirable chemical reactions that would "burn" the material and produce petroleum coke. The vacuum gas oil is sent to the catalytic cracking unit for further processing. The vacuum residua is sent to a coking unit for further processing or to fuel oil blending.
 The fractions from the crude and vacuum distillation units are then sent to fuel blending or other downstream processing units as shown in the following chart. The chart or flowsheet represents the simplest type of refinery which distills the crude oil into its natural components. Additional refining processes are used to increase the production of higher-valued products such as gasoline and diesel fuel and improve the quality of of the finished products.
The fractions from the crude and vacuum distillation units are then sent to fuel blending or other downstream processing units as shown in the following chart. The chart or flowsheet represents the simplest type of refinery which distills the crude oil into its natural components. Additional refining processes are used to increase the production of higher-valued products such as gasoline and diesel fuel and improve the quality of of the finished products.Catalytic Reforming
Catalytic reforming is used to improve the quality of naphtha from the crude distillation unit. The catalytic reforming unit uses a catalyst to allow the chemical reactions to take place under "reasonable" temperatures and pressure and "encourage" the desired hydrocarbons to be produced. The motivation for using catalytic reforming can be seen in the following table:
| Hydrocarbon | Hexane | Hexene | Cyclohexane | Benzene |
| Hydrocarbon Type | Paraffin | Olefin | Naphthene | Aromatic |
| Research Octane Number | 25 | 80 | 83 | 106 |
Therefore this process provides higher octane material to the gasoline pool to help meet the octane specifications on the gasoline. The process also produces hydrogen which is used to remove sulfur from refinery streams in the hydrotreating processes.
Catalytic Cracking
Catalytic cracking is a very important process in the modern refinery. The process allows the refiner to convert material that would normally be burned as fuel (vacuum gas oil) into gasoline and distillate (home heating oil and diesel fuel). One only need examine the price difference between residual fuel oil and gasoline to see why this is an attractive alternative. (For current prices see Gulf Coast Spot Product Prices).
This process breaks or cracks long chain hydrocarbons into smaller molecules in the naphtha and distillate boiling range to increase gasoline and diesel production. This process will yield 50-60% gasoline, 20-30% distillate and 30% butanes and lighter. If you do the math you will see that the volume of products is greater than the volume of the feed. This is because the long chain hydrocarbons are broken into smaller ones.
Alkylation and Isomerization
In the alkylation process, isobutane is reacted with either isobutylene or propylene to form complex paraffin isomers. the reactions take place in the presence of hydrofluoric or sulfuric acid catalysts. By combing these molecules the octane level of the paraffin isomer or alkylate is increased to around 93-96 octane. Refiners use this process to improve the octane level of the gasoline pool.
Light naphtha (90-190 oF.) can have its octane number improved by the use of an isomerization process to convert normal paraffins into their isomers. This results in an increase in octane number as evidenced by increase in normal pentane (62 octane) to iso-pentane (92 octane). The process uses a platinum catalyst. Like alkylation, this process improves the octane quality of the gasoline pool.
Hydrotreating
Hydrotreating is a process where a petroleum fraction is reacted with hydrogen for the purpose of removing impurities. The process is usually used to remove sulfur. Hydrotreating processes use hydrogen from the catalytic reformer or a hydrogen plant.
Product Blending
Product blending is where the different petroleum fractions are combined together to make the final product. The fractions are mixed so they meet the specifications discussed earlier. Each product has a specific recipe that calls for the proper mix of petroleum fractions. For example, in order to make gasoline, the refiner would mix naphtha, reformate, catalytic gasoline, alkylate and butane so that the mixture had the required octane number, vapor pressure, sulfur level and aromatics content. The process requires knowing these values for all of the components going into the blend. The recipes are developed using computer models.
Refinery Complexity
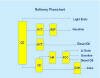 Refineries can range from simple topping plants with only a crude oil distillation tower to the more complex refinery shown in the flowsheet. This refinery includes reforming (REF), catalytic cracking (FCC) and coking (COK). Sulfur is removed using the hydrotreating (HYT) and hydrodesulfurization (HR) processes.
Refineries can range from simple topping plants with only a crude oil distillation tower to the more complex refinery shown in the flowsheet. This refinery includes reforming (REF), catalytic cracking (FCC) and coking (COK). Sulfur is removed using the hydrotreating (HYT) and hydrodesulfurization (HR) processes. Refinery product yields vary from different parts of the country as seen in the following chart:
U.S. Refinery Yields , Percentage of Crude Oil Charge
| Product | PADD I East Coast | PADD II Midwest | PADD III Southwest | PADD IV Rocky Mountains | PADD V West Coast | Total |
| LPGs | 3.2 | 3.5 | 5.1 | 1.6 | 2.8 | 4.1 |
| Gasoline | 44.6 | 48.4 | 41.6 | 47.3 | 45.2 | 44.2 |
| Jet Fuel | 5.7 | 6.3 | 9.4 | 4.8 | 17.5 | 9.6 |
| Distillate Fuel Oil | 29.5 | 30.0 | 28.3 | 31.6 | 21.6 | 27.8 |
| Residual Fuel Oil | 7.1 | 1.6 | 4.0 | 2.2 | 5.5 | 4.0 |
| Petroleum Coke | 3.3 | 4.3 | 5.9 | 4.6 | 6.0 | 5.3 |
| Asphalt & Road Oil | 5.1 | 5.3 | 1.2 | 6.1 | 1.4 | 2.7 |
| Petrochemical Feedstock | 1.1 | 1.0 | 3.7 | 0.0 | 0.1 | 2.2 |
| Other | 5.8 | 5.1 | 7.7 | 5.7 | 6.5 | 6.4 |
| Total | 105.4 | 105.5 | 106.9 | 103.9 | 106.6 | 106.3 |
[Source: EIA, Refinery Yield, 2008.]

Tidak ada komentar:
Posting Komentar